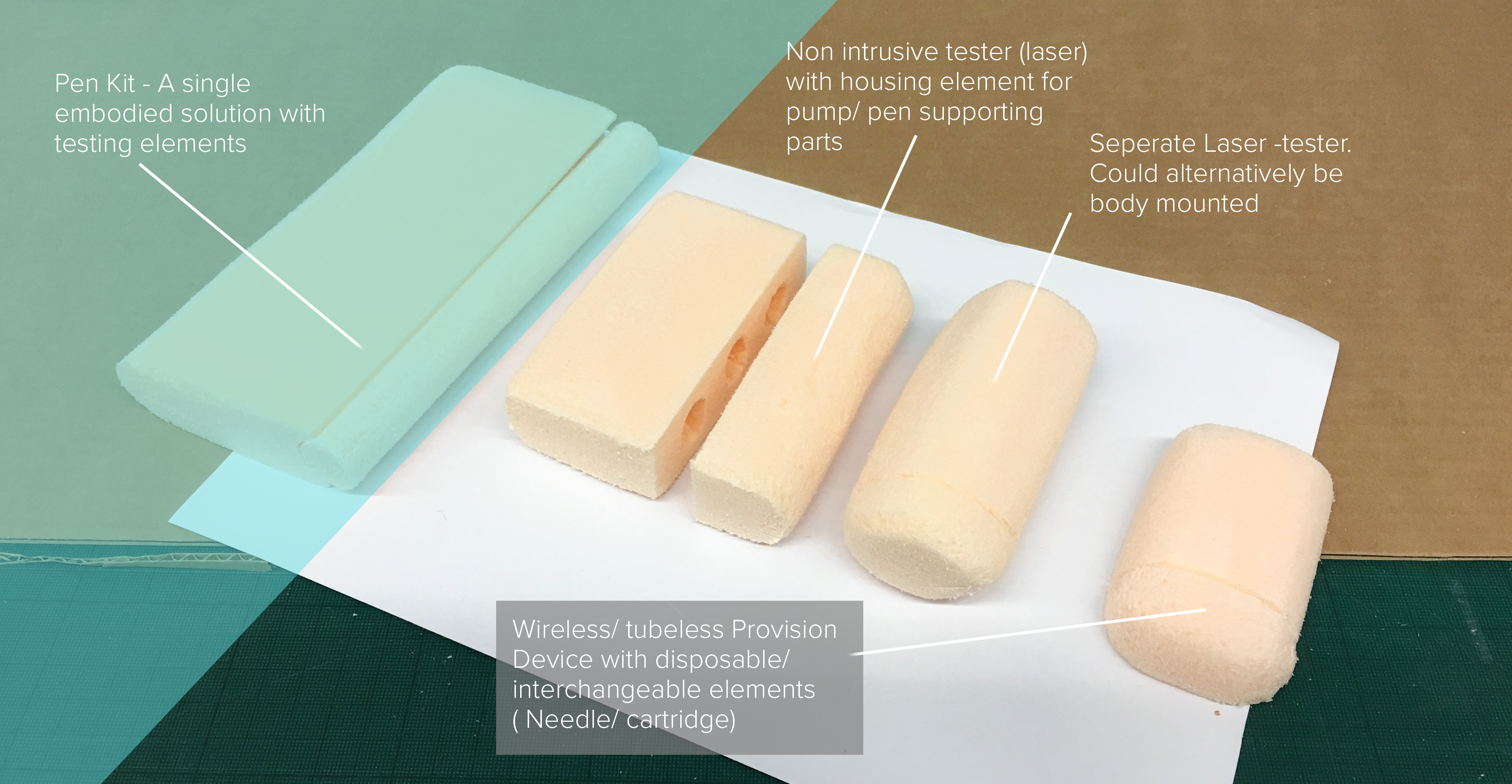
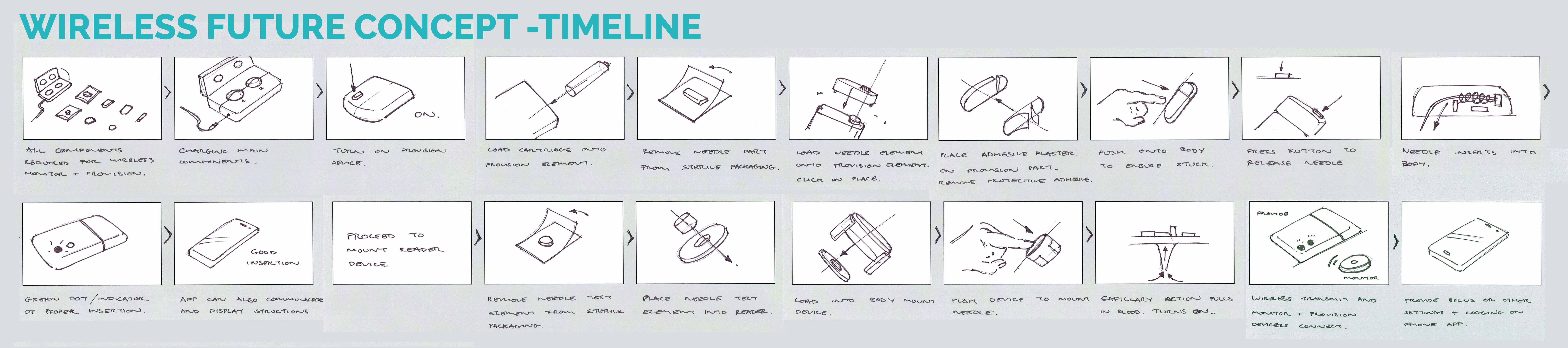
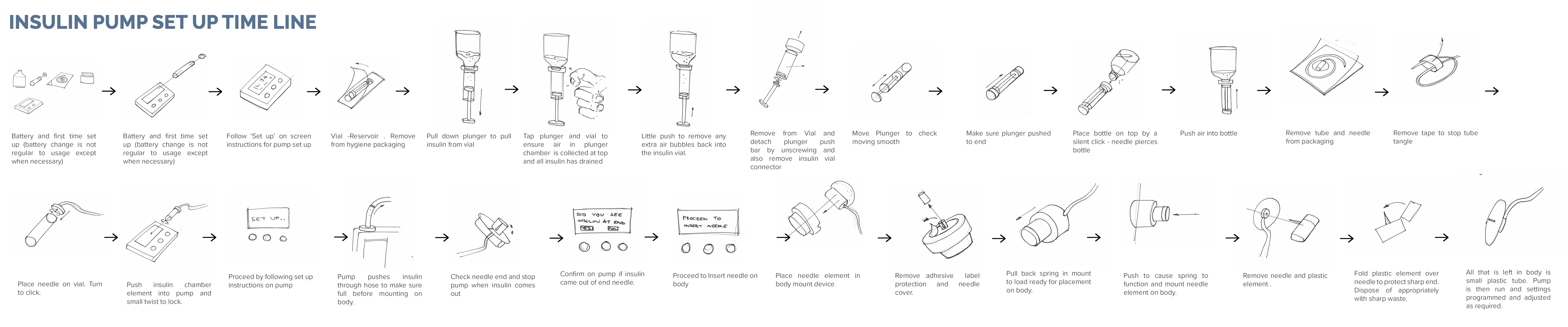
Very interesting and relevant lecture by Ian Hewitt entitled “Design for Medical and Well Being”
Ian discussed one of the most important considerations when designing medical products which is standards and classifications which are determined by Implant, intrusive and data recording.
There are 3 classes:
Class 1
- Low risk .. e.g. walking stick, wheel chair
Class 2
- Stringent Regulations
- Record data, Limited diagnosis
- E.g. Syringe, gloves, pregnancy Kit- self diagnostic
Class 3
- Highest category
- High Risk- complex devices
- Pace Maker, Ocular tweezers, Insulin Pen, Cannula.

Considerations:
- British Standards
- If it has a battery – needs to be able to be accessed later for recycling
Cleaning
- Surface finish
- Limitations
- How easy
Maintenance
- Is it externally serviced
- Parts
- Screws- easily accessible
- Parts based- disposable
Reuse
- Sterilised
- Collected
- Stored
- Re- distributed
Consider
- NHS may not likely adopt your project just due to the system , pricing range, competitors … “disruptive innovation”
- Labels
- Colours
Data recording
- Legality with holding data
- Has to be stored in a certain way
Diagnosis
- Safety
- Delivery of Information
Sharps
- Disposal
- Storage
- Contamination
- Replaceable
Materials
- Paint
- Material type (Allergies)
- Finishes
End User
- Age groups
- Different user perspectives
Consider constructing DARL and CRL logs as method for development
An incredibly relevant and useful lecture and I have discussed to have a meeting with Ian in the upcoming weeks to gain his thoughts and views on the project.
 Dr Rachel Clarke quoted a famous Dr William Osler “Just listen to your patient, he is telling you the diagnosis”.
Dr Rachel Clarke quoted a famous Dr William Osler “Just listen to your patient, he is telling you the diagnosis”.